

Gambogic Acid
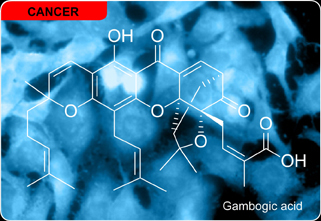
HeLa cells treated with gambogic acid under ambient light become fluorescent. Preliminary studies now indicate that once in a cell gambogic acid underoges a conjugate addition reaction with glutathione across its enone motif. This process leads to formation of amine-Michael adduct that reserves its thiol for protein glutathionylation. Using SDS PAGE gel analysis, we have observed the formation of disulfides between the amine-linked glutatione-gambogic acid conjugate and cysteine residues in proteins. In turn, this suggests that gambogic acid and related caged Garcinia xanthones could exert their biological action by regulating protein glutathionylation. When conducted under ambient light, proteins isolated from HeLa cells are irreversibly tagged and emits blue fluorescence. This labeling of proteins upon photoexcitation of glutathione adduct can be explained by considering insertion of a sulfur radical to a nearby amino acid. This program was terminated in 2011.
Copyright 2004-2012 | The Xenobe Research Institute | a California-based 501(c)(3) organization.