

Marinopyrroles
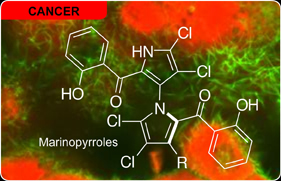
The marinopyrroles are a unique class of densely halogenated, axially chiral metabolites that contain an uncommon bispyrrole structure (read more here). We have begun studies on determining the mode of action of the marinopyrroles in tumor cells and bacteria in collaboration with the Fenical laboratory (Scripps Oceanographic Institute), Dorrestein laboratory (UC San Diego) and Madeleine Joullié (University of Pennsylvania).
Recently, we described the targeting of the marinopyrroles to actin by use of a dye transfer technique. The targeting of marinopyrrole A to actin was identified using a fluorescent dye transfer strategy. The process began by appending a carboxylic acid terminal tag to a phenol in the natural product. The resulting probe was then studied in live cells to verify that it maintained activity comparable to marinopyrrole A. Two-color fluorescence microscopy confirmed that both unlabeled and labeled materials share comparable uptake and subcellular localization in HCT-116 cells. Subsequent immunoprecipitation studies identified actin as a putative target in HCT-116 cells, a result that was validated by mass spectral, affinity, and activity analyses on purified samples of actin. Further data analyses indicated that the dye in the marinopyrrole probe was selectively transferred to a single residue K(115), an event that did not occur with related acyl phenols and reactive labels. In this study, the combination of cell, protein, and amino acid analysis arose from a single sample of material, thereby, suggesting a means to streamline and reduce material requirements involved in mode of action studies (read more here).
The total synthesis of marinopyrole A has been completed in the Nicolaou laboratory (read it now), Sarli laboratory (read it now), Li laboratory (read it now). Further biological studies now describe the utility of these materials Methicillin-Resistant Staphylococcus aureus (read it now) as well as Mcl-1 antagonists (read it now).
Copyright 2004-2012 | The Xenobe Research Institute | a California-based 501(c)(3) organization.